Dr. Ilo Leppik had a hunch.
Years ago, one of his epilepsy patients complained of painful side effects from her medication. No matter what he prescribed, the patient returned within two days with the same problems.
After checking with the head of the department and the Institutional Review Board (IRB), he prescribed her Cebocap.
Days after starting the new prescription, she was in the emergency room with the same set of side effects.
Unlike the slew of medications Leppik had prescribed before, Cebocap wasnâÄôt an epilepsy medication, but a placebo âÄî a âÄúsugar pill.âÄù
Just as Leppik had expected, his patientâÄôs side effects were the result of her own anxiety and not of the medication he had prescribed.
The idea that placebos can have positive or negative medical effects is nothing new.
But as the understanding of the connection between the mind and body has grown, so too has the interest in using placebos both in the examining room and in research.
Experts at the University of Minnesota have seen firsthand the effects sugar pills can have on patients.
Once commonplace in medicine, placebo use is highly contested by practicing physicians and is a topic of debate among researchers today.
In LeppikâÄôs case, it was the only instance in his 35-year career heâÄôd used a placebo.
âÄúThis is something we physicians donâÄôt do at all, except under very unique circumstances,âÄù he said.
Not âÄòmagic in the headâÄô
By definition, a placebo is any medical intervention âÄî most often a pill or surgery âÄî that doesnâÄôt actively treat the condition for which itâÄôs prescribed. Some placebos do have medical value, but not for the illness theyâÄôre prescribed to treat.
In one of its first appearances in medical history, a placebo was defined in QuincyâÄôs Lexicon-Medicum from 1811 as the name for âÄúany medicine adapted more to please than to benefit the patient.âÄù
Placebo itself means âÄúI shall please,âÄù in Latin.
The majority of the limited research into how placebos work has been focused on pain relief.
The two primary factors that lead to a placeboâÄôs effectiveness are expectation and conditioning, said Dr. John Grabowski, a University psychiatry professor.
A patient who receives a placebo from a doctor doesnâÄôt know the medication is inert and therefore expects it to be effective.
Combined with cues from the doctor that relief is coming soon âÄî phrases like âÄúYouâÄôll start feeling better in two daysâÄù âÄî a patientâÄôs expectation can have a profound impact on health, even if the pills had no medicinal properties.
If a patient takes aspirin for joint pain for four weeks and then unknowingly takes a placebo, that patient will more than likely feel the same relief, Grabowski said. ThatâÄôs conditioning at work.
The two factors combine to activate opioids in the brain, which decrease the perception of pain.
âÄúIt isnâÄôt some sort of magic in the head,âÄù Grabowski said.
Though the understanding of how the placebo effect works has evolved rapidly, much remains unstudied.
The percentage of people who are receptive to placebos and what makes a person receptive are still up for debate.
Based on their clinical research experience, Grabowski and Leppik estimated between 10 and 20 percent of patients show improvement when unknowingly given a placebo.
But Grabowski said the pain relief from a placebo is typically temporary.
âÄúIn the end, the response to a placebo pain medicine is probably never going to be as good as the response to morphine,âÄù he said.
If there were a grant to study the variables that influence placebo reception, Leppik said heâÄôd be on board.
âÄúI would really go after measuring things like hormone levels, adrenaline levels and things like that,âÄù he said.
âÄòNecessary deceptionâÄô no more
The use of placebos in the doctorâÄôs office is very controversial today, but that wasnâÄôt always the case.
Placebo prescriptions were commonplace before World War II.
Now, the use of placebos in clinical practice is generally denounced by medical organizations as a violation of trust between the doctor and patient.
Yet roughly half of American physicians said they regularly prescribe placebos in order to improve a patientâÄôs expectations, according to a 2008 survey funded by the National Center for Complementary and Alternative Medicine.
Few doctors in that survey said they had prescribed sugar pills or other inert drugs, but instead recommended over-the-counter treatments, vitamins or antibiotics. Physicians commonly described their placebo prescriptions as atypical solutions that might work for the patientâÄôs condition, according to the survey.
Of the 672 doctors surveyed, 62 percent said using placebos was ethical.
The Minnesota Board of Medical Practice has no concrete policies in place to address the use of placebos in medical practice, Executive Director Robert Leach said.
The board will respond to complaints that a doctor has inappropriately prescribed placebos and has the authority to discipline that physician. In his more than 20 years at MBMP, Leach could not recall a single complaint and said he has not disciplined a physician for such an offense.
âÄúI find it unconscionable,âÄù Grabowski said of the use of placebos in clinical medicine.
âÄúWhile one can take advantage of it, writing a prescription for a placebo âĦ it really troubles me.âÄù
Placebo use in research
The ethical code for physicians doing research with human subjects is the Declaration of Helsinki, a set of principles developed by the World Medical Association. Those guidelines have been revised six times since their adoption in 1964.
Under the umbrella of human research are clinical trials, in which researchers try to find new and more effective therapies âÄî often for illnesses that already have medications.
Placebos can only be used as a control in clinical trials if a proven medication for the illness doesnâÄôt already exist, according to the declaration. But it condones placebo use if it is âÄúnecessary to determine the efficacy or safety of an intervention and the patients who receive placebo âĦ will not be subject to any risk of serious or irreparable harm.âÄù
Researchers in clinical trials often welcome that exception with open arms.
Placebos are crucial to compare against the medication being tested in order to determine whether itâÄôs effective.
Pharmaceutical companies are often reluctant to test their new medicine against a similar existing medication, Grabowski said, because researchers then have to show the newer medicine is more effective than the old.
Research institutions like the University must have an IRB that reviews all research projects with human subjects before testing can begin.
One IRB requirement for clinical trials is that all subjects must be aware they may not actually receive medication, but a placebo.
Grabowski, who has run clinical trials for the past 25 years, said he tells his subjects their chances of receiving a placebo are like âÄúflipping a coin,âÄù and describes it as such in the consent form.
But the health of many who donâÄôt receive the medication will still improve. As soon as patients enroll in a clinical trial, they tend to do better, Grabowski said.
âÄúAlmost all of the project coordinators IâÄôve worked with come across [as] very caring to the research subjects,âÄù said Dr. S. Charles Schulz, head of the UniversityâÄôs Psychiatry Department and a veteran of clinical trials for anti-psychotic medications.
The trial environment and the process of working with attentive doctors create another positive effect, which makes it more difficult to determine how much of the improvement was triggered by the medication.
âÄúWhen people are getting a heavy dose of the therapy, that adds up,âÄù Grabowski said. âÄúYou end up not being able to show the medication effect, even though it is doing things that make it better for the patient.âÄù
Subjects, not patients
In each of his clinical trials, Schulz uses a standard introduction to his research subjects, thanking each person for enrolling in the trial. He tells them that their contribution will help determine whether the medication is fit to be used on patients in the future.
ThatâÄôs the key: The volunteers are contributing to research and not getting treatment like they might at a doctorâÄôs office. Each person is a research subject, not a patient.
Being honest and upfront with subjects about the research is a crucial ethical issue. But simply telling subjects they may receive a placebo may not be enough, said Jeff Kahn, director of the UniversityâÄôs Center for Bioethics.
âÄúPart of the challenge in the ethics of research is that âĦ [subjects] still wonâÄôt appreciate that they could be randomized into an arm that will receive the placebo,âÄù Kahn said.
No matter how open researchers are, people who enroll in clinical trials still want to improve their health. Any confusion is generally not due to misbehavior on the part of researchers, he said, but a challenge of the research environment in general.
âÄúItâÄôs a balancing act,âÄù Kahn said. âÄúWe donâÄôt want them to know if theyâÄôre getting placebo or not. We want them to know that itâÄôs possible.âÄù
Kahn is also concerned with âÄúsham surgeriesâÄù performed in clinical trials.
For example, in certain ParkinsonâÄôs disease research, researchers will perform surgery to implant certain drugs or medical devices in the brain. The placebo group in studies like that will go under the knife but unknowingly wonâÄôt receive the therapy being tested.
âÄúYouâÄôre now introducing risk of harm without any potential offsetting benefit to those individuals,âÄù Kahn said.
Those who undergo a sham surgery in trials will sometimes see an improvement in health, much like in drug trials.
There are several alternatives to using placebos as a control in clinical trials, but their use has become what Kahn calls the âÄúgold standardâÄù in comparative medicinal research.
âÄúIt shows the power of oneâÄôs desire to get better.âÄù
Daily Email Edition
Get MN Daily NEWS delivered to your inbox Monday through Friday!

Published April 26, 2024

Published April 26, 2024

Published April 26, 2024
Trending
Placebo ethics: real or perceived?
For years, medical experts have been divided over the ways in which placebo use is fair — if at all.
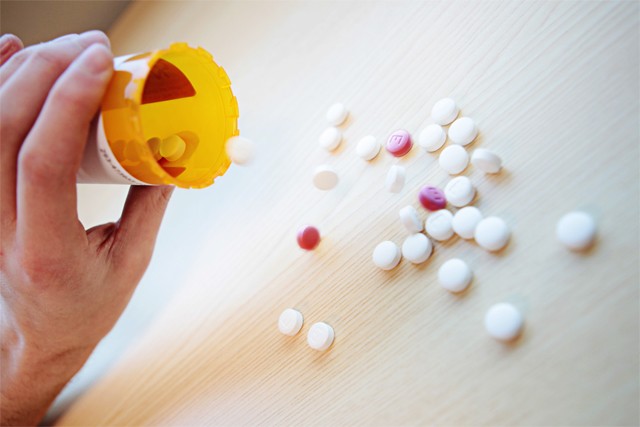
Image by Mark Vancleave
Placebos, or “sugar pills,” are used both in clinical trials and as prescriptions. Experts have raised ethical questions of a violation of trust between a doctor and patient.
by Kyle Potter
Published March 23, 2011
Leave a Comment
More to Discover
Advertisement
The Minnesota Daily • © 2024 The Minnesota Daily • Privacy Policy • FLEX Pro WordPress Theme by SNO • Log in